ETHICAL ISSUES
THE ANTIDEPRESSANT ISSUE

- Anti-depressants have been commly used to treat depression. According to a report by the Center for Disease Control and Prevention (CDC) adult use of antidepressants has grown tremendously since 1988 with " Ten precent of women 18 and older and 4 precent of men now take antidepressants" (CDC p.1)
-With the growth of demand for antidepressants comes the need for testing of medications. The FDA requires that new treatments have greater effectiveness than placebo. Opponents to the use of placebo state that their use in testing violates the "best proven diagnostic and therapeutic method" of the orginial Heisinki Declaration of 1964. It is also believed that the use of placebo may cause patients to suffer due to not having their medication. One of the arguments by those that are pro-placebo is that the concern should be the "risk to participants" and if there is no risk danger to the participants then the useage of the placebo is justified.
-Kirsch, Moore, Scoboria, and Nicholls (2002) conclued that the recent published clinical trials reveal that "75 precent of the response to antidepressants is duplicated by placebo". Due to the severity of the side effects of antidepressants the placebo effect has brought about an ethical issue.
-Should patients that have adverse side effects to antidepressant be given placebos? The issue here is that this my invole deception which would challenge the requirement to be open and honest to patients.
-With the growth of demand for antidepressants comes the need for testing of medications. The FDA requires that new treatments have greater effectiveness than placebo. Opponents to the use of placebo state that their use in testing violates the "best proven diagnostic and therapeutic method" of the orginial Heisinki Declaration of 1964. It is also believed that the use of placebo may cause patients to suffer due to not having their medication. One of the arguments by those that are pro-placebo is that the concern should be the "risk to participants" and if there is no risk danger to the participants then the useage of the placebo is justified.
-Kirsch, Moore, Scoboria, and Nicholls (2002) conclued that the recent published clinical trials reveal that "75 precent of the response to antidepressants is duplicated by placebo". Due to the severity of the side effects of antidepressants the placebo effect has brought about an ethical issue.
-Should patients that have adverse side effects to antidepressant be given placebos? The issue here is that this my invole deception which would challenge the requirement to be open and honest to patients.
Figure. 1 Chart
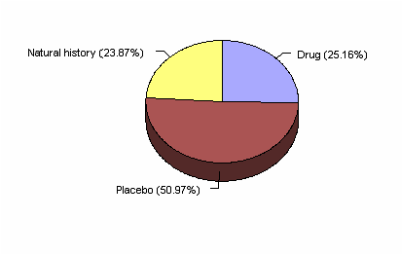
This chart is data collected from the response to antidepressant medication.
According to a study by Kirsch, and Sapirtein (1998) one quarter of the drug response from the group was cause by the medication, one quarter is caused by nonspecific factors, and the remaining one half is a placebo effect.
TREATMENT-RESISTANT DEPRESSION ISSUE

-Studies show that "more than one thrid of the patients treated for depression will become treatment resistant.
-Many patients are misdiagnosised TRD due to the absence of a clear criteria for TRD.
-Littrell and Lacasse (2012) found that atypical antipsychotic were being used more to treat patients with TRD. The concern with these durgs are the side effects which can include brain volume reduction, weight gain, motor problems and heart problems.
-Many patients are misdiagnosised TRD due to the absence of a clear criteria for TRD.
-Littrell and Lacasse (2012) found that atypical antipsychotic were being used more to treat patients with TRD. The concern with these durgs are the side effects which can include brain volume reduction, weight gain, motor problems and heart problems.
